 It arranges of the elements in order of increasing atomic number. Francium, however, is a laboratory-produced element and only minute quantities have been made, so for all practical purposes, the most reactive metal is cesium. The periodic table is used as a predictive tool. Nonmetal reactivity increases from left to right and decreases down groups. nonmetallic characteristics increases from left to right and decreases down groups. These trends explain the periodicity observed in the elemental properties of atomic radius, ionization energy, electron affinity, and electronegativity. The most reactive metal on the periodic table is francium. Metal reactivity decreases from left to right across periods and increases down groups. This happens because the number of filled principal energy levels (which shield the outermost electrons from attraction to the nucleus) increases downward within each group. For example: Lithium (Li) reacts quickly with water. The reactions of fluorine with most other elements range from vigorous to explosive only O 2, N 2, and Kr react slowly. The specific arrangement of Group 1 elements on the periodic table allows us to make predictions about their reactivity. Second, moving down a column in the periodic table, the outermost electrons become less tightly bound to the nucleus. Fluorine is the most reactive element in the periodic table, forming compounds with every other element except helium, neon, and argon. As this happens, the electrons of the outermost shell experience increasingly strong nuclear attraction, so the electrons become closer to the nucleus and more tightly bound to it. Atoms of an atomic number may have varying numbers of neutrons (isotopes) and electrons (ions), yet remain the same chemical element. First, electrons are added one at a time moving from left to right across a period. The periodic table lists elements by atomic number, which is the number of protons in every atom of that element. 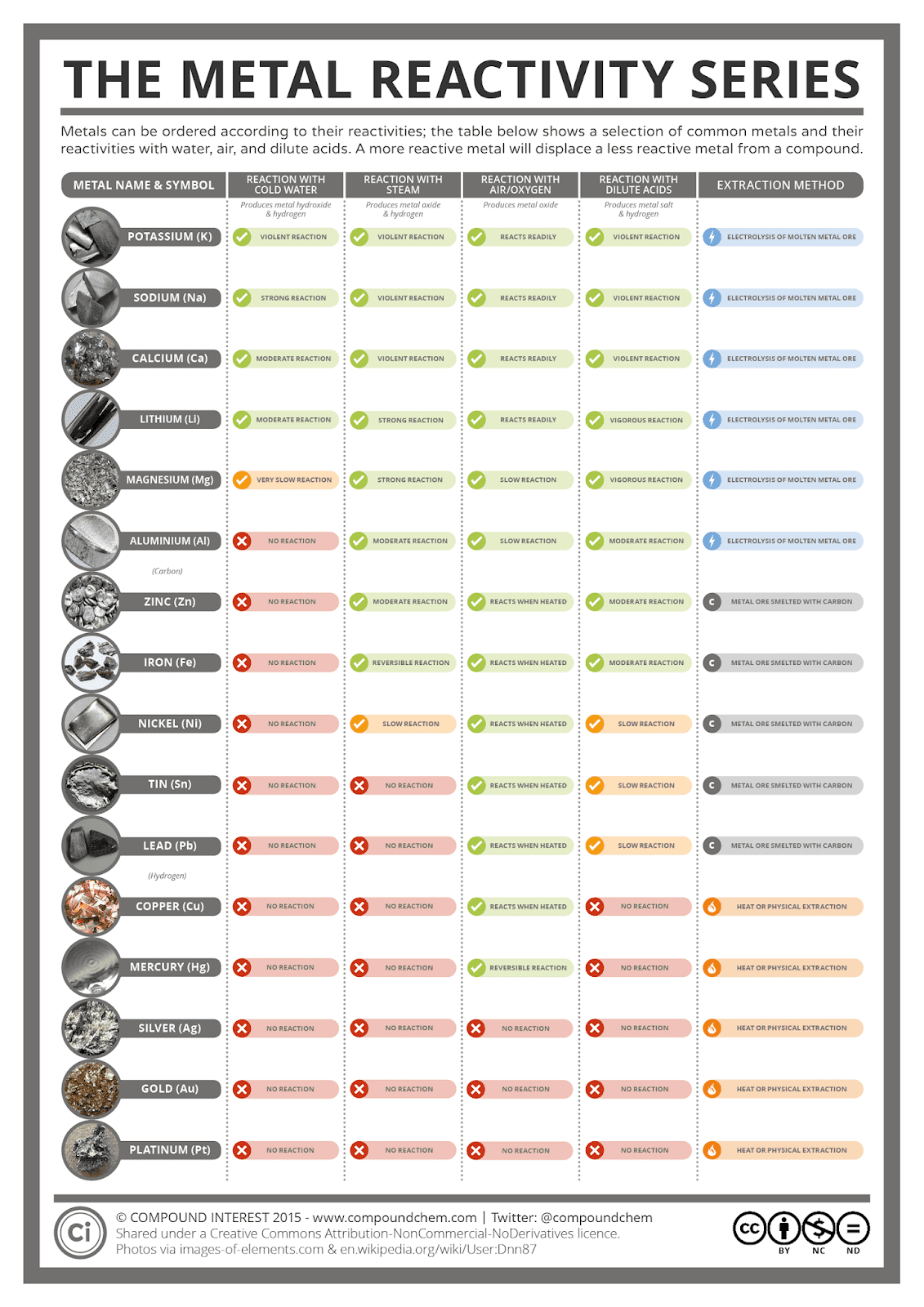
In addition to this activity, there are two other important trends. Stable octets are seen in the inert gases, or noble gases, of Group VIII of the periodic table. Elements tend to gain or lose valence electrons to achieve stable octet formation. Understand the patterns in formula of compounds. Play this game to learn about lots of different elements in the periodic table. These trends can be predicted merely by examing the periodic table and can be explained and understood by analyzing the electron configurations of the elements. Fluorine is a highly reactive element, and form fluorides with the majority of other elements. The periodic table arranges the elements by periodic properties, which are recurring trends in physical and chemical characteristics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
